Development of CO2-derived ion-conductive polymer materials for safer all-solid-state lithium batteries
October 17, 2017
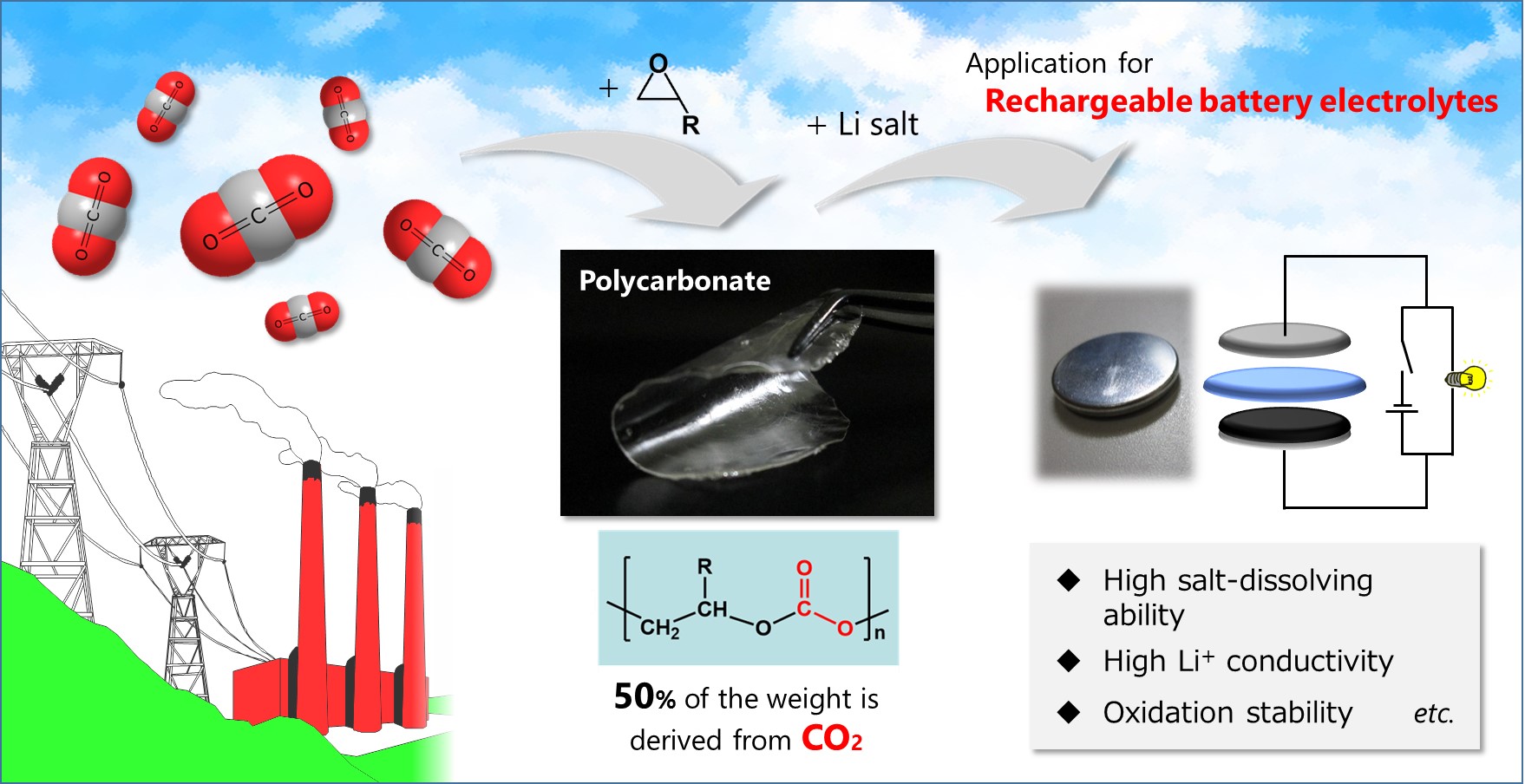
Associate Professor Yoichi Tominaga (Division of Applied Chemistry, Institute of Engineering, Tokyo University of Agriculture and Technology), Kento Kimura (Graduate School of Bio-Applications and Systems Engineering, Tokyo University of Agriculture and Technology) and their research group discovered that a solid polymer electrolyte based on poly(ethylene carbonate) (PEC), which is derived from CO2, is suitable for use in high-voltage all-solid-state lithium rechargeable batteries on the basis of an unusual ion-conductive mechanism. Lithium batteries using solid polymer electrolytes are safer than current systems based on volatile and flammable organic liquid electrolytes. The present work is expected to facilitate the furthering of electric vehicles and renewable energies, and will contribute to realizing a sustainable energy use. This research was presented on September 20 in the 66th Symposium on Macromolecules held at Ehime University, Matsuyama, Japan. A part of this research has been published on May 27, 2016 in The Journal of Physical Chemistry C (http://pubs.acs.org/doi/abs/10.1021/acs.jpcc.6b03277).
Solid polymer electrolytes (SPEs), which are mixtures of polar polymers and metal salts, are soft ion-conductive materials, and are particularly suitable for lithium rechargeable batteries. SPEs are promising materials as alternatives to volatile and flammable liquid electrolytes. So far most studies have been directed to polyethers, such as poly(ethylene oxide) (PEO). Practical use of polyether electrolytes, however, is difficult because of low ionic conductivity and low compatibility with battery electrode materials. An alternative strategy is therefore necessary.
The present study looked at the ion-conductive behavior of an SPE based on poly(ethylene carbonate) (PEC). The ionic conductivity of PEC electrolyte increases with increasing salt concentration, and the electrolytes have high lithium transference numbers. This is an unusual tendency, which is opposite to that of the conventional polyether electrolyte. The mechanism of the unusual behavior was not fully understood. In the present study the group utilized spectroscopic measurements to study the correlation between the structural feature and the ionic conduction. The results confirmed that a loose solvation structure of the dissolved salt and a highly aggregated ionic structure give rise to the unusual performance. Furthermore, the resulting highly concentrated electrolytes show extraordinary oxidation tolerance and prevention effect of metal corrosion reaction, on the basis of the highly concentrated character. These results give a new insight into the development of SPEs having high conductivity, high lithium transference number, and high electrochemical stability based on the highly concentrated character, and also into a possibility of CO2 utilization as a raw carbon source of functional polymer materials.
Acknowledgment
This work was supported by JSPS KAKENHI Grant Number 25288095, 16H04199, 16J04407, and the Specially Promoted Research for Innovative Next Generation Batteries of the Advanced Low Carbon Technology Research and Development Program (ALCA-SPRING) from the Japan Science and Technology Agency (JST), Japan.
Related Information
TUAT research factors (English): http://www.rd.tuat.ac.jp/en/activities/factors/index.html
Researcher information (Japanese):Yoichi Tominaga
Press release (Japanese): 二酸化炭素を原料とするイオン伝導性高分子材料~安全な固体リチウム二次電池開発に貢献~(PDF:368KB)
Contact
Yoichi Tominaga
Associate Professor, Division of Applied Chemistry, Institute of Engineering,
Tokyo University of Agriculture and Technology
Tel +81-42-388-7058
